Compounds constituted of a porphyrin core bound to one β-cyclodextrin unit were synthesized and characterized by MALDI-TOF, NMR spectroscopy, gel permeation chromatography and UV-VIS spectrophotometry. We have synthesized initially 5,10,15-tri[p-(9-methoxy-triethylenoxy)-phenyl]-20-[p-(n-bromo-n-ane-1-oxy)-phenyl]-porphyrin by reaction between 5,10,15-tri[p-(9-methoxy-triethylenoxy)-phenyl]-20-(p-hydroxyphenyl)-porphyrin and Br(CH2)nBr.
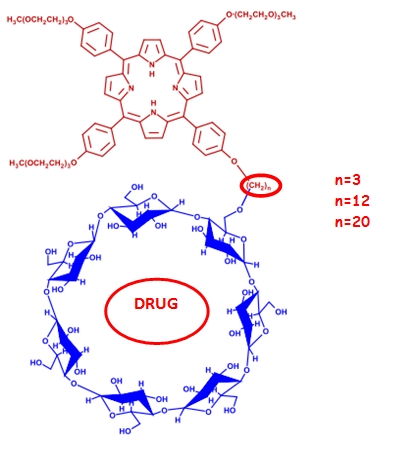
The conjugates were obtained finally by reaction between 5,10,15-tri[p-(9-methoxy-triethylenoxy)-phenyl]-20-[p-(n-bromo-n-ano-1-oxy)-phenyl]-porphyrin[H2-P(TEG-ME)3-O(CH2)n Br] and sodium β-cyclodextrine-6-alcoholate. Compounds with n=3, 12, 20 were synthesized. These coniugates combine the capability of cyclodextrin unit to host poorly soluble drugs (such as many anticancer drugs), with the photosensitizing and "tumor targeting" properties of porphyrins.
The possibility to apply simultaneously two therapeutic modes, (photodynamic therapy and chemotherapy), allows to obtain a higher therapeutic effect respect to the single approach for treatment of cancer. In addition the possibility of changing metal ion, substituents linked to the meso porphyrin positions , length and nature of the "bridge" between cyclodextrin and porphyrin unit, allows to optimize the system for the therapeutic requirements.
People to contact: Daniele Vitalini - Emanuela Spina
PON01_00074 (DIATEME -15/09/2011-30/05/2015).